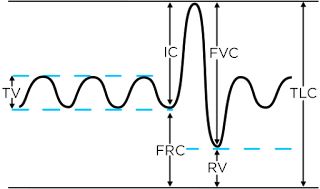
In clinical medicine, spirometry continues to dominate as the pulmonary function test of choice. This is despite the issues which limit its utility, largely patient compliance and the limited inferences which can be made regarding the mechanical properties of the respiratory system. Alternatively, the forced oscillation technique (FOT), utilized by the
flexiVent preclinically, is well-known for its rich description of the mechanical properties of the respiratory system. However, despite becoming the gold-standard in preclinical research, the approach has yet to make significant in-roads into the clinical domain, especially in the low frequency range (0-2 Hz) that gives access to the peripheral lung, the area most sensitive to pathological changes.
In ventilated patients FOT solutions do exist, however free-breathing patients present obstacles as accurate measurements of the respiratory system rely on precise measurements of pressure and flow. Potential solutions to this problem have been trialed, for instance training patients to perform voluntary apnea
1 or by triggering an apneic event in infants using the Hering-Breuer reflex
2.
A recent doctoral thesis by Dr. Hannes Maes, under the supervision of Prof. Gerd Vandersteen at Vrije Universiteit Brussel (Free University of Brussels), titled,
Low Frequency Forced Oscillation Technique in Clinical Practice, proposes an innovative solution to overcome confounding breathing frequencies, as well as a comprehensive review of the field
3.
Dr. Maes presents a novel fan-based setup specifically designed for FOT measurements between 0-5 Hz in free-breathing subjects
1 Pressure signals (multi-sine wave) are superimposed on the patients normal tidal breathing by controlling two fans (one pushing and one pulling air). Compensatory signals are also applied to the fans to ensure a predefined power spectrum and to suppress the nonlinear influences of the equipment or the subject’s breathing, along with post-measurement modeling techniques.
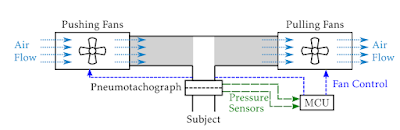 |
| Schematic of fan-based FOT measurement device for free-breathing subjects |
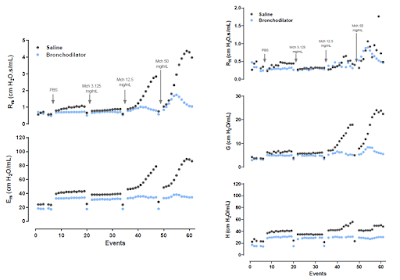
Results from a clinical trial on 60 subjects, that included healthy individuals as well as asthmatic and COPD patients, are also presented and, in the end, Dr. Maes offers a new framework for the time-varying behavior of the respiratory system during spontaneous breathing.
We congratulate Dr. Maes on this achievement and very interesting approach. This elegant work contributes towards the advances required to ultimately enable routine provision of the low-frequency FOT in patient populations and for clinicians to benefit from its diagnostic capacity.
For more information on the work of Dr. Maes and access to the full manuscript, please click
here.
1. Hantos, Z., Daróczy, B., Suki, B., Galgóczy, G. & Csendes, T. Forced oscillatory impedance of the respiratory system at low frequencies. J. Appl. Physiol. 60, 123–32 (1986).
2. Hall, G. L., Hantos, Z., Wildhaber, J. H. & Sly, P. D. Contribution of nasal pathways to low frequency respiratory impedance in infants. Thorax 57, 396–9 (2002).
3. Maes, H. Low Frequency Forced Oscillation Technique in Clinical Practice. (Vrije Universiteit Brussel, Belgium, 2017).